 
In nature, uranium isotopes are typically found in radioactive equilibrium (i.e. The activity concentration arising solely from the decay of the uranium isotopes (U-234, U-235 and U-238) found in natural uranium is 25.4 Bq per mg. An activity of one becquerel (Bq) means that on average one disintegration takes place every second. 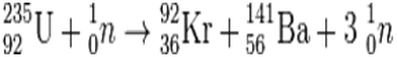
It is expressed in the table in becquerels (Bq) per milligram (1 milligram, mg, = 0.001 grams). The specific activity is the activity per unit mass of a particular radionuclide and is used as a measure of how radioactive a radionuclide is. The half life of a radioactive isotope is the time taken for it to decay to half of its original amount of radioactivity. The table below shows the fraction by weight of the three isotopes in any quantity of natural uranium, their half lives, and specific activity. Other isotopes that cannot be found in natural uranium are U-232, U-233, U-236 and U-237. In its natural state, it consists of three isotopes (U-234, U-235 and U-238). The International Atomic Energy Agency (IAEA) defines uranium as a Low Specific Activity material. It is so dense a small 10-centimetre cube would weigh 20 kilograms. Like tungsten it is very dense, about 19 grams per cubic centimetre, 70% more dense than lead. In its pure form it is a silver-coloured heavy metal, similar to lead, cadmium and tungsten. While different, the two processes have an important role in the past, present and future of energy creation.Uranium (chemical symbol U) is a naturally occurring radioactive element. Fission is the splitting of a heavy, unstable nucleus into two lighter nuclei, and fusion is the process where two light nuclei combine together releasing vast amounts of energy. However, research continues into ways to better harness the power of fusion, but research is in experimental stages, as scientists continue to work on controlling nuclear fusion in an effort to make a fusion reactor to produce electricity.īoth fission and fusion are nuclear reactions that produce energy, but the processes are very different. Fusion reactions are not easily controlled, and it is expensive to create the needed conditions for a fusion reaction. These benefits are countered by the difficulty in harnessing fusion. Fusion offers an appealing opportunity, since fusion creates less radioactive material than fission and has a nearly unlimited fuel supply. Some scientists believe there are opportunities to do so. While fission is used in nuclear power reactors since it can be controlled, fusion is not yet utilized to produce power. The high-speed neutrons that are ejected become projectiles that initiate other fission reactions, or chain reactions. This resulting energy is then used to heat water in nuclear reactors and ultimately produces electricity. This splits the target nucleus and breaks it down into two smaller isotopes (the fission products), three high-speed neutrons, and a large amount of energy. During the process, a neutron is accelerated and strikes the target nucleus, which in the majority of nuclear power reactors today is Uranium-235. These neutrons are accelerated and then slammed into the unstable isotope, causing it to fission, or break into smaller particles. 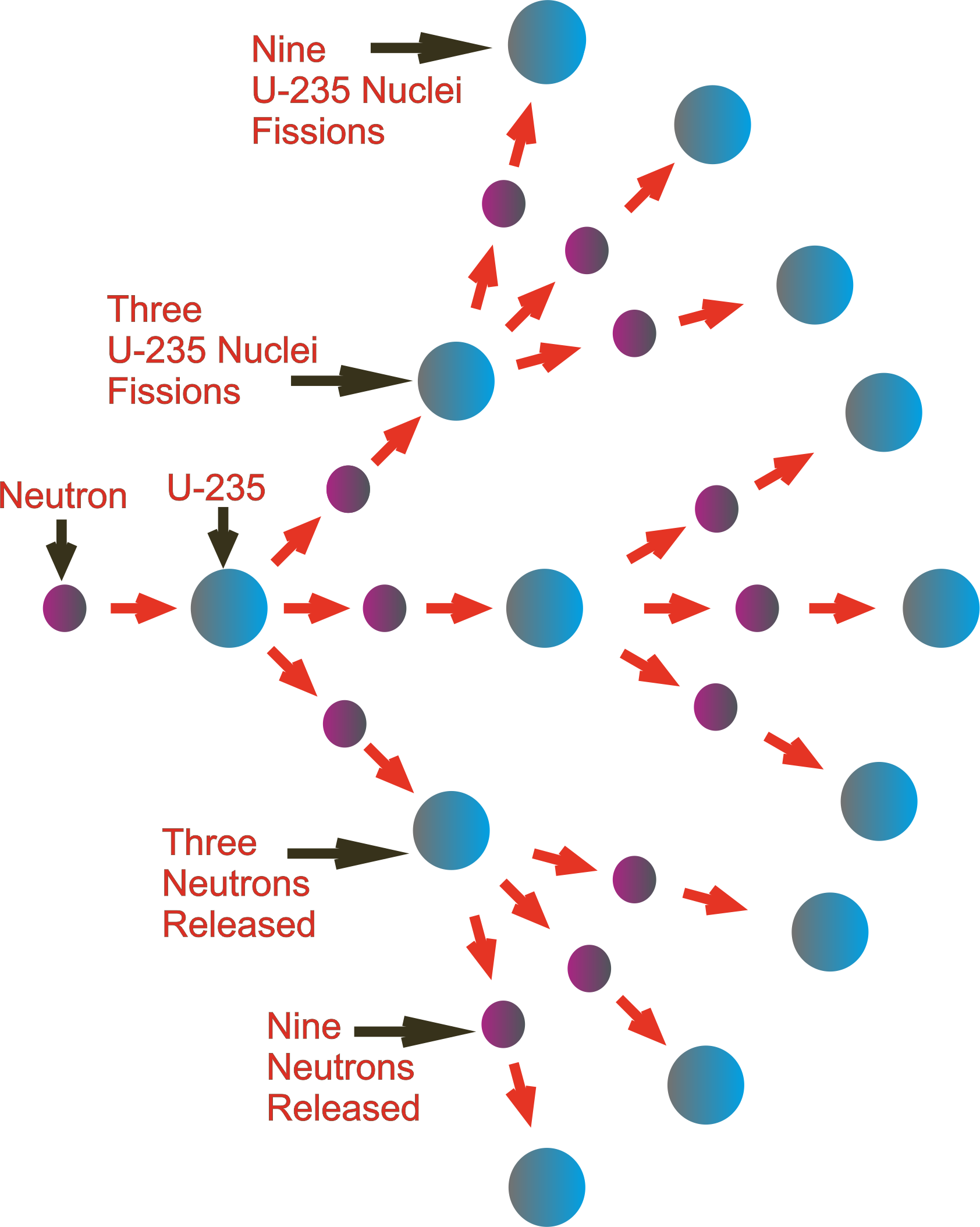
Fission takes place when a large, somewhatunstable isotope (atoms with the same number of protons but different number of neutrons) is bombarded by high-speed particles, usually neutrons. Just as cell’s divide, in fission an atom splits into smaller particles. Both fission and fusion alter atoms to create energy, but what is the difference between the two?įission, a term coined by scientists LIse Meitner and Otto Frisch, is named after the term “binary fission” in biology to describe cell division. The foundation of nuclear energy is harnessing the power of atoms by splitting apart, a process called fission, or combining them, called fusion. Inside the sun, fusion reactions take place at very high temperatures and enormous gravitational pressures. Look up during the day to see one of the most powerful examples of a nuclear reactor: the sun. Inside the sun, fusion reactions take place at very high temperatures and enormous gravitational pressures 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
